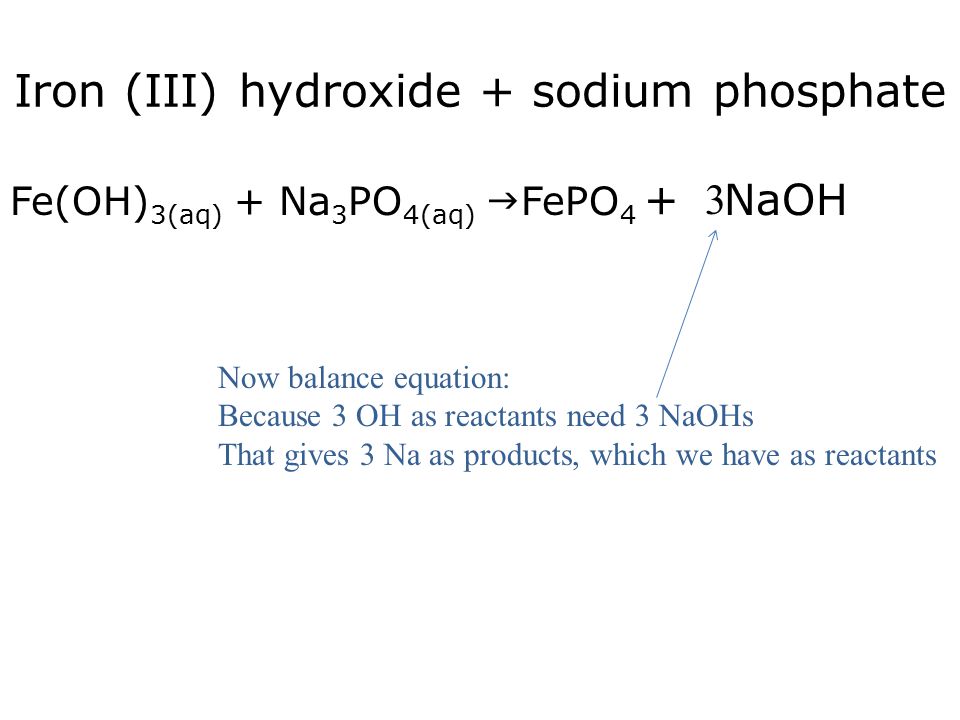
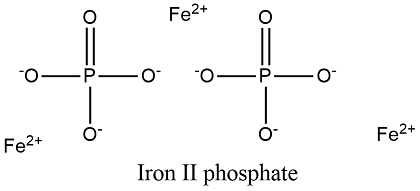
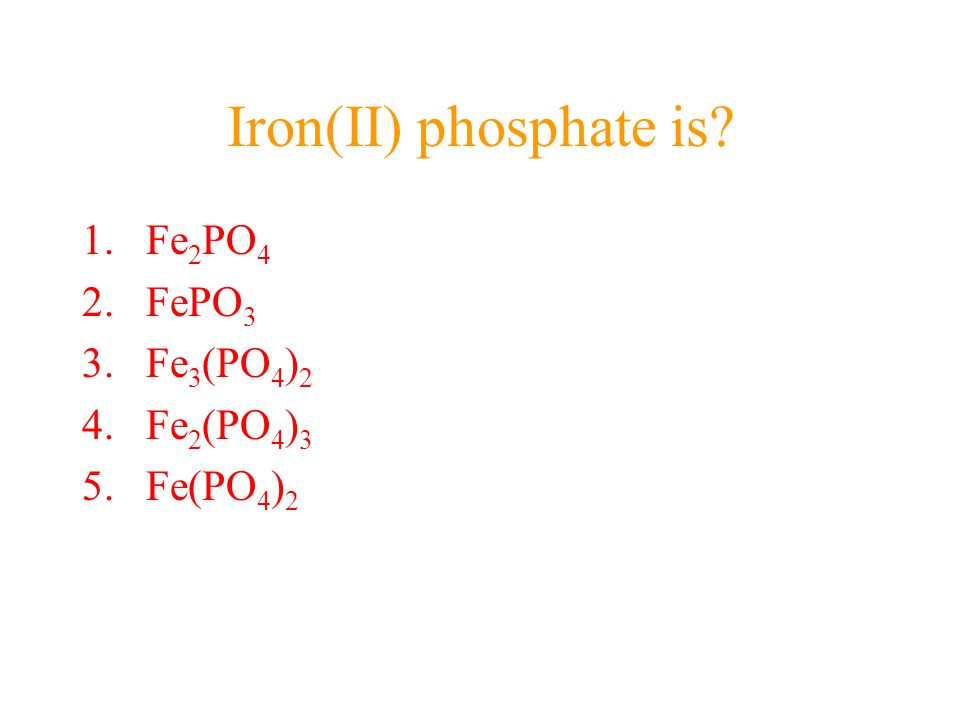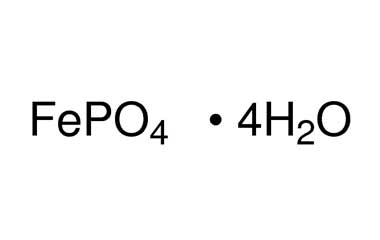SOLVED: Write the balanced molecular equation, including phases, for the reaction of aqueous sodium phosphate with aqueous iron(II) nitrate: molecular equation: Na3PO4(aq) + Fe(NO3)2(aq) -> Fe3(PO4)2(s) + 2NaNO3(aq)
Iron(III) Phosphate Lithium Iron Phosphate Iron(II) Sulfate PNG, Clipart, Angle, Bicarbonate, Brand, Carbonate, Chemical

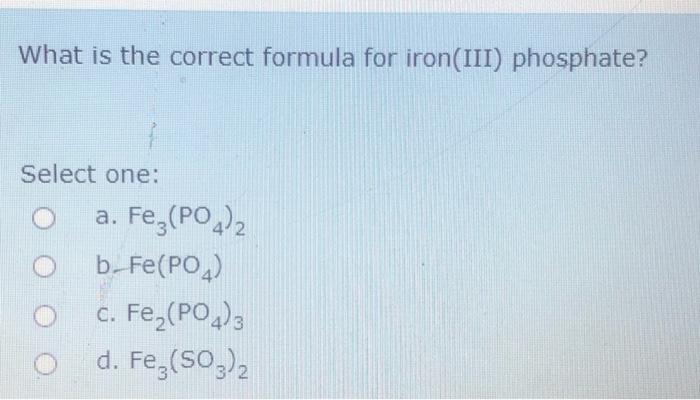
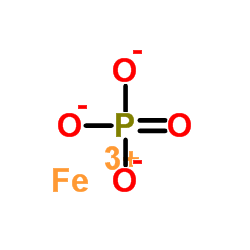
10045-86-0 | Ferric Phosphate | Iron Phosphate; Escargo!; Ferramol; Ferric Orthophosphate; Iron Orthophosphate; Iron Phosphate; Iron(III) Phosphate; Sluggo; | FePO₄ | TRC

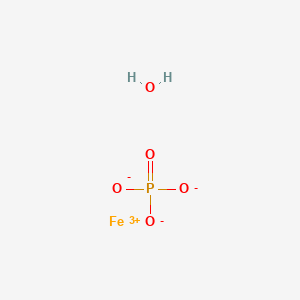
SOLVED: Q6: The correct name of "FePO4·4H2O" is A) Iron(II) phosphate tetrahydrate B) Iron(III) phosphate tetrahydrate C) Iron(II) phosphate hydrate D) Iron(II) phosphate tetrahydrate